RESEARCHER SPOTLIGHT
MARK HEIMAN, PHD
Mark Heiman
Mark Heiman co-invented the drug Lanreotide, which is used to treat pituitary adenomas, spent more than 20 years at Eli Lilly & Co where he was Chief Science Officer of obesity drug discovery, and has recently credited the ICBI for providing a unique environment that led to his latest research advances.
When he was walking among the graduate and medical students at Louisiana State University Medical School in the late 1970s, his classmates couldn’t have known that one of their own would earn his PhD in medical physiology (and later his postdoctoral training at IU Medical School in neuroendocrinology) and go on to author over 100 scientific papers, including his seminal work on the roles of leptin and ghrelin in regulating energy balance. But if they’ve been paying attention over the years, they know that, at an age when most of us are slowing down, Mark Heiman is still going strong and continuing to achieve impressive results in the process.
Heiman is a Fellow of the Obesity Society and a member of The Endocrine Society where he plays an advisory role in the Entrepreneurship special interest group. He has served on editorial boards for Endocrinology, Molecular Metabolism and the Journal of Hepatology & Gastroenterology, as well as on NIH study sections.
Although he professes to be retired, you may still see him walking the halls of the ICBI; he is the Chief Science Officer for Scioto Biosciences, an ICBI company since 2018. Heiman has a special interest in the gastrointestinal microbiome and its role in health and disease.

Current Studies
Mark Heiman and the Scioto Biosciences team have been using their knowledge of the bi-directional interaction among the brain, gut, and gut microbiome by administering live bacterial therapeutics. The composition of the 100 trillion gut microorganisms plays a role in health and disease, and the opportunity to use microorganisms in the gut to develop effective therapies for neurological and digestive disorders has been gaining traction. The gut informs the central nervous system of its status through molecules derived from the microbiota, neuroactive substances, and gut hormones that travel to the brain through the enteric nervous system, vagus nerve, and circulatory or immune systems. Once in the brain receives these substances, it can generate an appropriate response to that physiological or pathological state. This gut-microbiota-brain axis impacts neurodevelopment. Heiman and his team are harnessing the vagus nerve pathway to areas of the brain responsible for social interaction and reward in people with autism spectrum disorder (ASD).
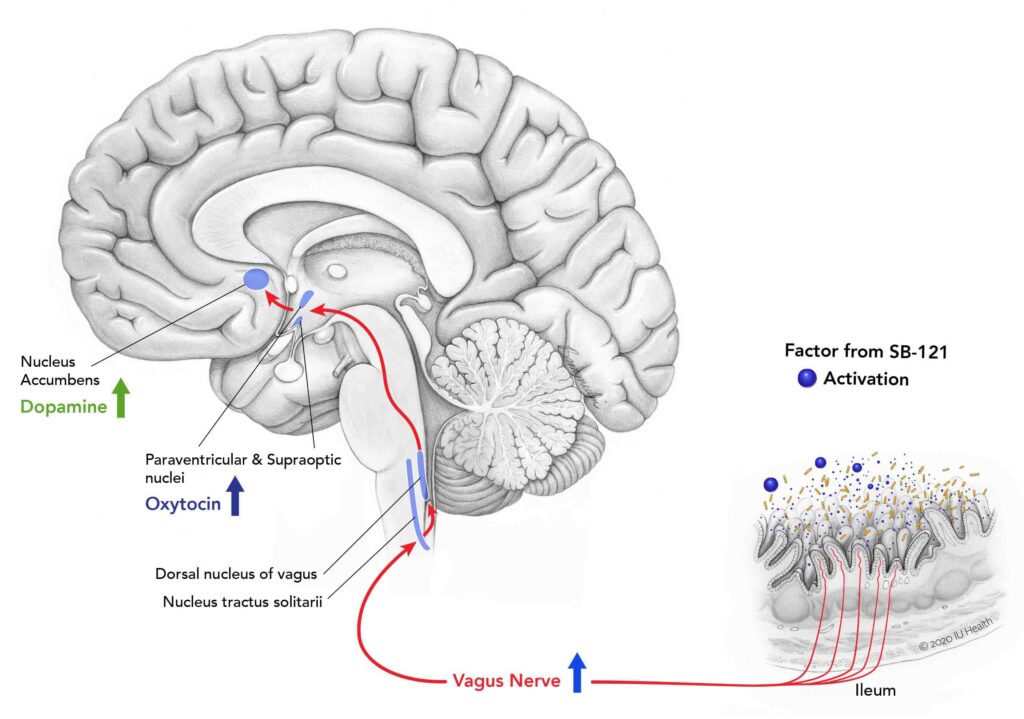
Their leading proprietary candidate is SB-121, which is a combination of the probiotic bacterium Lactobacillus reuteri (L. reuteri), dextran microparticles that bind and activate the bacteria, and maltose that provides the fuel for activation.
The job of SB-121 is to colonize the L. reuteri in the ileum, which in turn protects against opportunistic pathogenic bacteria and stimulates release ofoxytocin and dopamine.
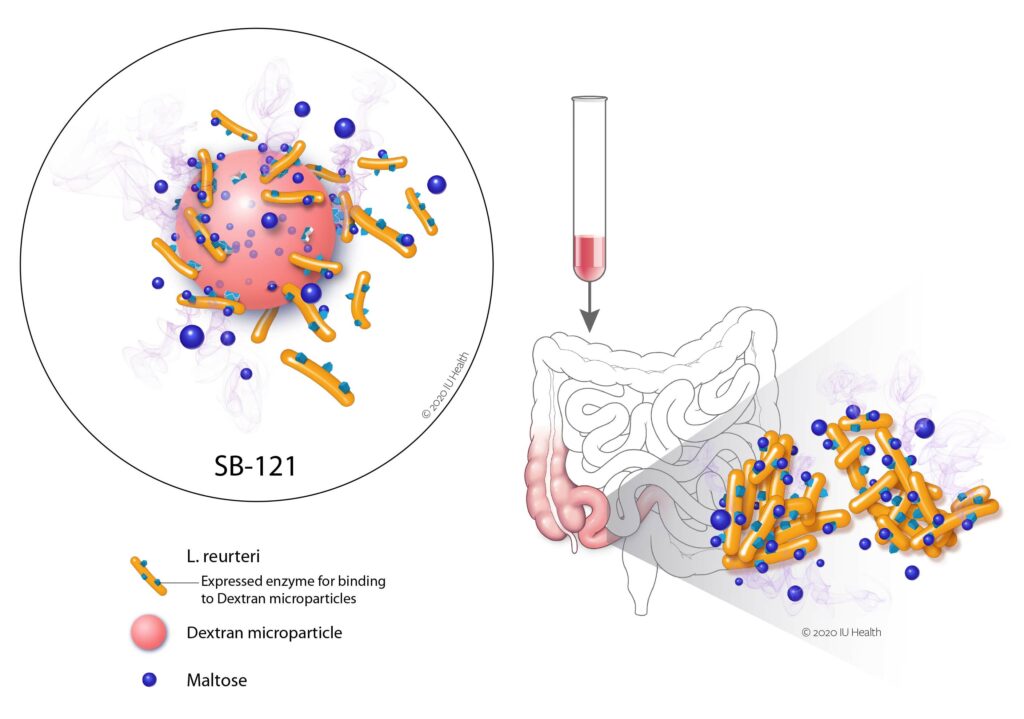
The live therapeutic bacteria are delivered already in a biofilm state in the SB-121 oral suspension, enabling the beneficial bacteria to persist and colonize the outer mucous layer of the intestines better than if the probiotic were ingested without the activated biofilm-forming state.
Scioto Biosciences’ latest publication describes their double-blind, placebo-controlled Phase 1b Clinical Trial in patients with Autism Spectrum Disorder (ASD), which showed that SB-121 was safe, well-tolerated, and most importantly, demonstrated treatment-related improvements for patients.
The ICBI Experience
There are several advantages that ICBI offers to startup companies, such as access to existing instruments and supplies that are too expensive for beginning budgets. The ICBI culture is one of sharing and collaboration that Heiman states is rare when comparing to incubators where he has worked in the past. A most important advantage of being a scientist at ICBI, in Mark Heiman’s opinion, is access to the IU Health vivarium. Not all life science incubators have this feature, and in Heiman’s case, the convenient access to his study animals meant that he was able to observe them frequently, providing observations that he would not have made otherwise. For Heiman, this was crucial in his recent work. In preliminary research that led to Scioto’s Phase 1b study, Heiman observed the spontaneous weaning of rat pups who were given SB-121. Rat pups normally transition from breast feeding to consumption of solid food about day 21. Rat pups who were treated with SB-121 were more curious, more exploratory, and transitioned to solid rat chow four days earlier than pups of the same age but treated with placebo. This observation suggested that SB-121 may have a role in neurodevelopment of social behavior. Scioto Biosciences then directed their focus on SB-121 for treatment of social disorders such as ASD.
Another benefit of the ICBI incubator for growing companies includes access to a medical illustrator employed by IU Health, a convenient resource that was tapped by Scioto Biosciences to help explain their research to investors—for those times when a picture really is worth a thousand words. (Illustration examples are seen above in the Current Studies section.)
Additionally, as Heiman revealed, the equipment in common research areas within the ICBI is another plus for company startups trying to stretch their capital, allowing for a more strategic use of company resources.
And finally, the sense of community and being able to exchange scientific as well as business pearls should not be underestimated.

