RESEARCHER SPOTLIGHT: PART 2
MARK HEIMAN, PHD
SCIOTO BIOSCIENCES
Current Studies
Mark Heiman and the Scioto Biosciences team have been using their knowledge of the bi-directional interaction among the brain, gut, and gut microbiome by administering live bacterial therapeutics. The composition of the 100 trillion gut microorganisms plays a role in health and disease, and the opportunity to use microorganisms in the gut to develop effective therapies for neurological and digestive disorders has been gaining traction. The gut informs the central nervous system of its status through molecules derived from the microbiota, neuroactive substances, and gut hormones that travel to the brain through the enteric nervous system, vagus nerve, and circulatory or immune systems. Once in the brain receives these substances, it can generate an appropriate response to that physiological or pathological state. This gut-microbiota-brain axis impacts neurodevelopment. Heiman and his team are harnessing the vagus nerve pathway to areas of the brain responsible for social interaction and reward in people with autism spectrum disorder (ASD).
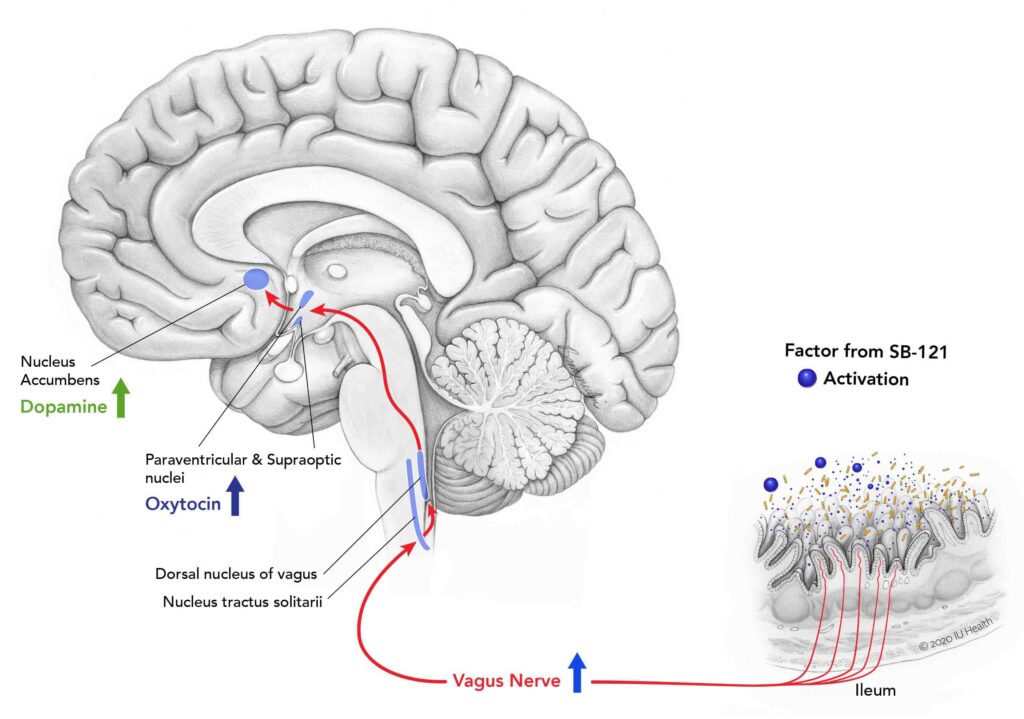
Their leading proprietary candidate is SB-121, which is a combination of the probiotic bacterium Lactobacillus reuteri (L. reuteri), dextran microparticles that bind and activate the bacteria, and maltose that provides the fuel for activation.
The job of SB-121 is to colonize the L. reuteri in the ileum, which in turn protects against opportunistic pathogenic bacteria and stimulates release ofoxytocin and dopamine.
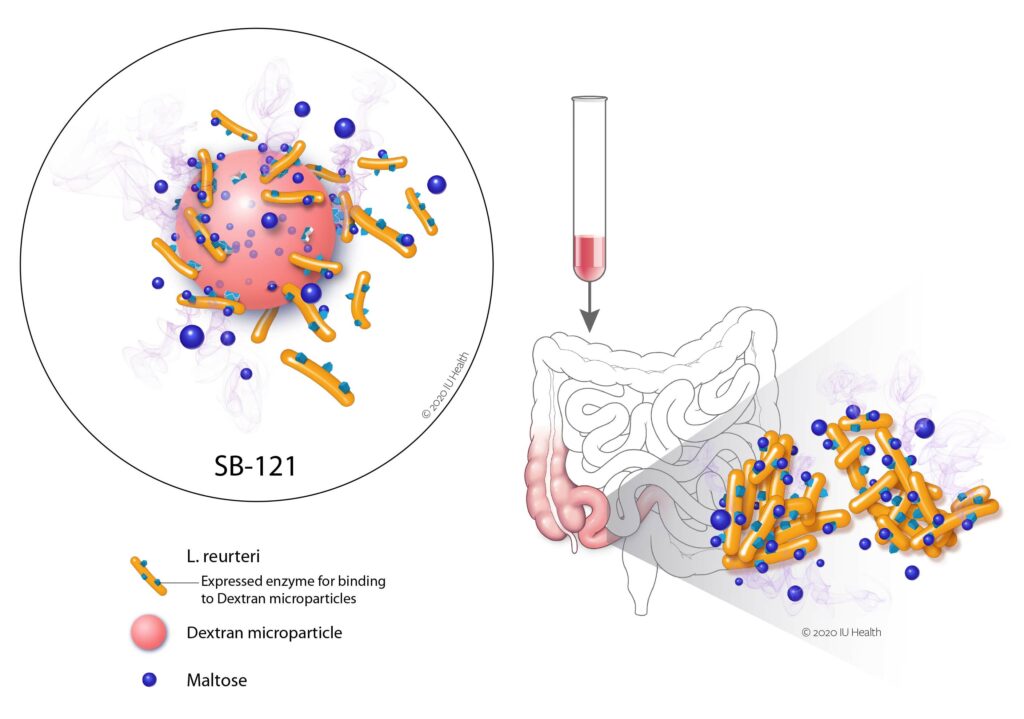
The live therapeutic bacteria are delivered already in a biofilm state in the SB-121 oral suspension, enabling the beneficial bacteria to persist and colonize the outer mucous layer of the intestines better than if the probiotic were ingested without the activated biofilm-forming state.
Scioto Biosciences’ latest publication describes their double-blind, placebo-controlled Phase 1b Clinical Trial in patients with Autism Spectrum Disorder (ASD), which showed that SB-121 was safe, well-tolerated, and most importantly, demonstrated treatment-related improvements for patients.
